导图社区 Host cell I and II
- 39
- 0
- 0
- 举报
Host cell I and II
Host cell I and II思维导图:包含Cellular homeostasis,Proteasome - antigen presentation or protein recycling ,These quality control measures generally lead to the degradation of mis-folded or foreign proteins等等
编辑于2022-05-10 13:44:24- B cell Immunodeficiency
这是一篇关于B cell Immunodeficiency的思维导图,其内容包括X—linked agammaglobulinemia(XLA),Hyper Igm syndrome等四个方面的内容
- NK Cells
这是一篇关于Natrual Killer Cells免疫学 墨尔本大学的思维导图,对于NK Cells感兴趣的小伙伴可以收藏起来。
- Bacteria Pathogenesis
Bacteria Pathogenesis思维导图:包含Normal Microbiota,Difference between Pathogens &Normal microbiota,Enter the body,Colonisation of the Host,Invasion of the host cell等等
Host cell I and II
社区模板帮助中心,点此进入>>
- B cell Immunodeficiency
这是一篇关于B cell Immunodeficiency的思维导图,其内容包括X—linked agammaglobulinemia(XLA),Hyper Igm syndrome等四个方面的内容
- NK Cells
这是一篇关于Natrual Killer Cells免疫学 墨尔本大学的思维导图,对于NK Cells感兴趣的小伙伴可以收藏起来。
- Bacteria Pathogenesis
Bacteria Pathogenesis思维导图:包含Normal Microbiota,Difference between Pathogens &Normal microbiota,Enter the body,Colonisation of the Host,Invasion of the host cell等等
- 相似推荐
- 大纲
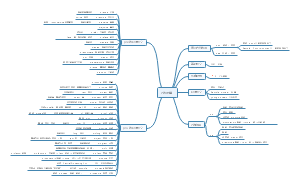
Host cell I and II
Intracellular transport
Mediating transport
Movement within lipid bilayer: clustering of receptors and signalling proteins facilitates by distinct lipid composition
Vesicular transport : mediated via distinct proteins “coat” the vesicles and target them to the appropriate location.
Cytoskeletal transport: mediated via microbules in a bi-directional manner
Microtubules and microtubule motor proteins
Kinesin drives transport from ER to PM
Dynein drives transport from Pm to ER
Pox virus utilisation of the cytoskeleton: moves from ER to PM via microtubules
Exit cells via actin
Transferring Cargo via membrane fusion
SNARE proteins mediated contact and fusion ‘mixing’ of vesicle membrane with target membrane
V-SNARE : vesicle snare
t-SNARE: target SNARE
Glycosylation in Golgi Apparatus
Plays a vital role in protein localisation, function and reognition
Secretory pathway
Used to move material aroun the cell
Various locations, including the PM and outside the cell
Proteins destined for the PM, endosomes/ lysosomes or secretion
Synthesised by the RER
Contain specific amino acid sequence (motif) which target them to lumen of RER
Proteins may be glucocylated while passing through the ER
Then bud from ER in vesicles
ER vesicles travels to cis face of Golgi Apparatus, then proceeds to trans face
Sometimes gaining additional modifications during this time which target proteins to the final location
Can do forward - antrograde. Can do backward - retrograde
Location of the motifs
Motif
Recognised by adaptor proteins embedded in the organelles membrane or by associated adaptor proteins
Each has different composition and property
Some have defined location within the proteins coding sequence whilst other do not
ER signals sequence: will embedded in ER; only found in N-terminus
Import into the ER
This motif is a stretch of 12-16 hydrophobic amino acids at the N-terminus flanked by basic ‘charged’ residues
These are termed ‘ signal sequence or signal peptide’
Once translocation into the ER has occurred the signal peptide is removed by the enzyme signal peptidase
Nuclear localisation motif (NLS): in the middle part of the amino acids sequences
Import into the ER
If the protein contains additional hydrophobic regions, these will embed the proteins within the membrane
Multiple ‘membrane spanning’ or ‘transmembrane’ domains may exist within a protein
These proteins then become tightly associated with the membrane
The greater the hydrophobicity of the protein the greater the affinity for membranes
ER retention motif (KDEL/KXXX): only found in C-terminus
Export from the ER
COPI: retrograde (back to ER)
Luminal proteins contain the motif - KDEL at their C terminus
Recognised by the KDEL receptor that interacts with COPI
Membrane-bound proteins contain a - KXXX motif within their cytoplasmic c-terminus (interacts directly with COPI)
COPII: anterograde (coats the vesicle leave the ER)
Cellular homeostasis
All cell in our body have innate mechanisms of quality control
Ensures that all proteins and constituents are functional
Ensures that any abuse (toxins or infection) are monitored and reacted to - digestion, degradation of molecules, particle, foreign microbes
These quality control measures generally lead to the degradation of mis-folded or foreign proteins
Unfold protein response - translation restriction and cellular stasis
Protein maturation requires protein chaperones to correctly fold the proteins
With in ER
Some of the chaperones will recognise misfiled proteins, export these proteins from the ER
These unfold proteins become ubiquitinated and sent to the proteasome for degradation
Proteasome - antigen presentation or protein recycling
Correct folding of proteins is critical in targeting them to their intended location
Unfold and mis-fold proteins are:
Secreted into the cytosol
Target for destruction by ubiquitination: attachment of several ubiquitin molecules
Degraded by the proteasome:
Small peptides and aa are produces: recycling of amino acid
Ubiquitin and degraded produces are released back into the cytosol
Energy dependent process
Proteasome produce peptides for antigen presentation during immune response (CTL and MHC I
Ubiquitin - proteasome pathway
A complex of three protein E1,2,3, which together constitute a ubiquitin ligase, recognise a protein destined for degradation and takes this proteins with a chain of ubiquitin termed polyubiquitylation
Following polyubiquitylation, the protein is carried to a proteasome where is a de-ubiquitylated and degraded into oligopeptides
Degraded further into amino acids or used for antigen presentation by the immune system
A specialised proteasome is used to process proteins for antigen presentation
Ubiquitin itself is a relatively short protein of 76 aa residues whose aa sequence and structure are almost totally conserved among all eukaryotic cell
Endocytic Pathway
Endocytosis observed in all Euk. Cell
Regular occurrence for recycling membrane molecules
Can be mechanisms for: cell feeding, digestion, degradation of molecules. Particles, foreign microbes
Bring extracellular material into the cell
Pinocytosis
Intakes of solutes
Phagocytosis
Intakes of particles via engulfment by membrane protrusion
Receptor-mediated endocytosis
Intakes of specific molecules which bind to receptors on the cell surface
Cathrin-mediated Endocytosis
Mediated via vesicles coated in the molecules - clathrin
Consist of three polypeptides chains that forms a triskelion
Triskelion assemble together to from a ‘cage-like’ structure (vesicles)
Generally occurs once a ligand engages with its receptor and promotes association between the cargo molecules, the receptor, adaptor proteins and clathrin to drive the reaction
The GTPase dynamin is then required to pinch the vesicle from the membrane
Once formed the clathrin-coated vesicles is then transported to the endosome
Endosomes aid in the process of endocytosis
Move material for degradation in lysosomes
Different classes
Early, late and recycling
Virus entry
Viruses utilise the process of endocytosis to gain entry into cells
Essentially they act as a ligand and bind to a cell surface receptor to promote the accumulation of clathrin and endocytosis
Lysosomes
Bud from the Golgi apparatus
Maturation pathway: early endosome - late endosome - lysosome
Functions
Involved in intracellular digestion / degradation pathway
Acid pH
Contains
Enzymes
Reactive Oxygen intermediates
Autophagy - recycling of organelles
Second degradation pathway in lysosome
A process where the cell degrades/ digest its own constituents and recycle them (expect nucleus)
Autophagosome - enclosures of an organelle with a double membrane
Dependent on a large number of cellular proteins and occurs in a highly regulated manner
Autophagesome fuses with a lysosome and the internal contents are degraded by hydrolyase and thus released
Appears to be anti-pathogen response but also exploited by pathogens
Plays a key role in the detection of pathogens and subsequent activation of the immune response
Can stimulate our innate and adaptive response
Deliver antigen to MHC I and II
Stimulate cytokine production
Conversely many pathogens manipulate the antophagy resopnse
Can enable replication
Can enable immune evasion