导图社区 Classification of matte
- 33
- 0
- 0
- 举报
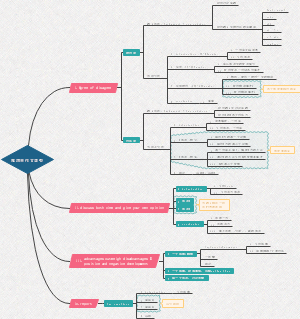
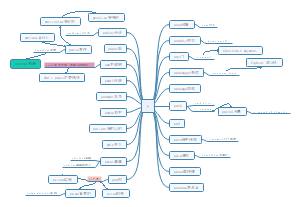
Classification of matte
Classification of matter:The cations generated when the electrolyte ionizesare all H+ compounds,Any of several distinct elements formed from the
编辑于2022-09-26 11:22:28- 相似推荐
- 大纲
Classification of matter
classification
Tree classification
A method of classifying substances according to their composition
Cross classification
A method of classifying the same substance according to different classification criteria
Oxide
Acid oxide
An oxide that reacts with a base to form salt and water
Basic oxide
An oxide that reacts with a acid to form salt and water
Amphoteric oxide
Eg. Iron oxide
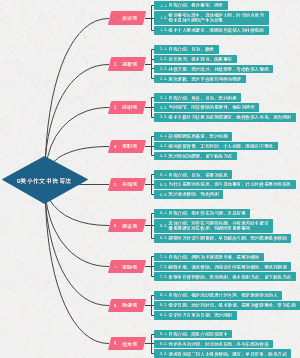
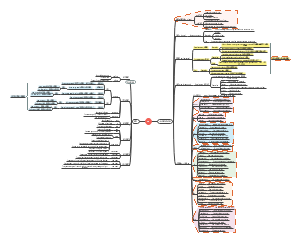
Equation of chemistry
Properties of oxygen
Magnesium burns in the air
Iron burns in oxygen
Hydrogen burns in the air
Red phosphorus burns in the air
Water in nature
Electrolysis of water (experiment to explore the composition of water)
Quicklime dissolves in water
Carbon dioxide is soluble in water
Law of conservation of mass
Iron reacts with copper sulfate solution
Reduction of copper oxide by oxygen
Carbon and carbon oxides
Reduction of copper oxide by charcoal
Marble reacts with dilute hydrochloric acid
Calcination of limestone at high temperature
Reaction of lime water with carbon dioxide (test for carbon dioxide)
Carbon monoxide reduces copper oxide
Flammability of carbon monoxide
Sodium carbonate and dilute hydrochloric acid (Fire extinguisher principle)
Fuel and its utilization
Methane burns in the air
Alcohol burns in the air
Hydrogen burns in the air
Metal
Zinc and dilute sulfuric acid (hydrogen made in the laboratory)
Iron and dilute sulfuric acid
Reaction of iron with a solution of copper sulfate
The principle of iron treatment and training
Acids, bases, salts
Reaction of ferric chloride with dilute hydrochloric acid
Reaction of hydrochloric acid with caustic soda
Reaction of marble with dilute hydrochloric acid (carbon dioxide prepared in laboratory)
Sodium carbonate reacts with dilute hydrochloric acid
Sodium carbonate and dilute hydrochloric acid (to test for deterioration of sodium hydroxide)
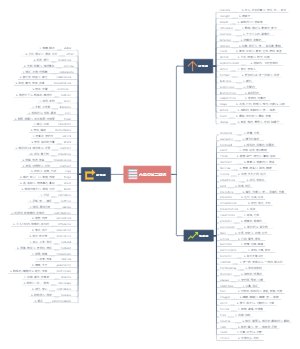
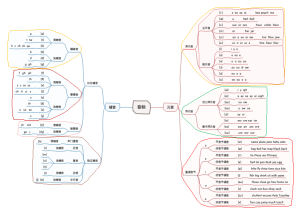
Chemical formula and valence
concept
A representation of the composition of matter as a combination of symbols and numbers
There's only one chemical formula for a pure substance, and there's only one chemical formula for a pure substance
Meaning of chemical formula
Water
Water is a substance
Water is made up of hydrogen and oxygen
It's a molecule of water
Represents a water molecule consisting of two hydrogen atoms and one oxygen atom
A chemical formula preceded by a number (a meaning)
2H2O:Two molecules of water
2He:That's two helium atoms
The writing of chemical formulas
elemental
Metal elemental
Solid nonmetallic element
Noble gas elemental
Gaseous nonmetallic elemental
Element symbol + number is pronounced as: X gas
Compound (two elements)
Writing: gold helium before, oxygen after, atom number lower right
Pronunciation: Read from back to front, add in the middle
valence
definition
The number of atoms that combine with each other
The application of valency
Denote the valence of the elements in the chemical formula
Write ion symbols according to the elemental valence shoe
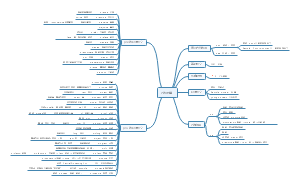
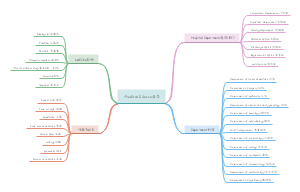
Electrolytes and non-electrolytes
The electrolyte
Strong electrolyte
Weak electrolyte
The electrolyte
Non metallic oxide
Sulfur dioxide
Nitrogen dioxide
Carbon dioxide
Gaseous oxide
methane
ammonia
Most organic matter
sucrose
alcohol
The difference between electrolyte and non-electrolyte
nature
Electrolytes can ionize, non-electrolytes cannot ionize
Compound of origin
Electrolytes are ionic or covalent compounds and non-electrolytes are covalent compounds
form
Hydrated ions or water and molecules
The electrical conductivity
The determinants
It depends on the concentration of ions, not how many ions there are
How a substance behaves in an aqueous solution
Water ionization
characteristic
Extremely weak, with few ions in the water, and almost no electricity
Endothermic: the degree of ionization increases with rising temperature
Salt
When ionized, cation (or ammonia ion) and acid ion compounds containing metals are formed
Normal salt
Sodium sulfate
Potassium sulfide
Acid salt
Sodium bisulfate
Potassium bicarbonate
Basic salt
Basic copper carbonate
alkali
solubility
soluble
Sodium hydroxide
Potassium hydroxide
The undissolved
Ferrous hydroxide
Iron hydroxide
Magnesium hydroxide
The degree of ionization
Strong
Sodium hydroxide
Potassium hydroxide
Barium hydroxide
Weak
Monohydrate and ammonia
Acid
The cations generated when the electrolyte ionizes are all H+ compounds
The degree of ionization
Strong
Sulfuric acid
Hydrochloric acid
Nitric acid
Weak
carbonic acid
Acetic acid
Allotrope
Any of several distinct elements formed from the same element