导图社区 Q1What is light
- 15
- 0
- 0
- 举报
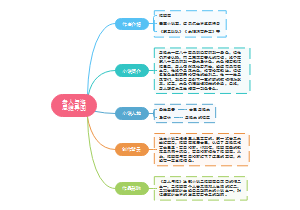
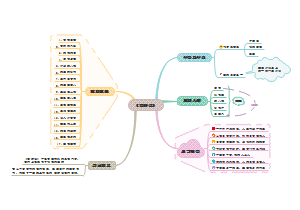
Q1What is light
这是一个关于Q1.What is light的思维导图,光是一种可以被人眼探测到的电磁波。电磁波可以有很宽的频率范围,从无线电波频率到伽马波频率。我们称这一范围的电磁波为电磁频谱。
编辑于2024-04-01 14:37:16- 光
- 证明光既是波又是粒子
- 相似推荐
- 大纲
Q1.What is light
Light is an electromagnetic wave that can be detected by the human eye. Electromagnetic waves can have a wide range of frequencies, from radio wave frequencies to gamma wave frequencies. We call this range of electromagnetic waves the electromagnetic spectrum. Because the human eye can detect only a small range of electromagnetic wave frequencies, visible light makes up only a small portion of the electromagnetic spectrum.
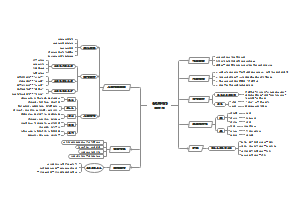
Christiaan Huygens
Time:1690s
Huygens theory of light fluctuations
Basic concept:This theory holds that light is not composed of particles, but is a wave phenomenon that travels through space, like water waves and sound waves. According to wave theory, the propagation speed of light waves will be different in different media, which explains the refraction, reflection and diffraction of light in different substances.
Optical experiment:Huygens supported his wave theory with a series of optical experiments. Among them, he is most famous for his research on the diffraction phenomenon of light. He found that when light passed through a small hole or around an obstacle, it produced a pronounced diffraction phenomenon, which could not be explained by the particle theory. In addition, Huygens also experimentally studied the interference phenomenon of light, which further confirmed the volatility of light.
Wave theory verification experiment:To test his wave theory, Huygens devised a series of experiments, the most famous of which was the double-slit interference experiment. Through this experiment, he observed that when light passes through two slits, it creates alternating interference streaks on the screen. This phenomenon is one of the strongest evidence for the wave theory, because it proves that light waves overlap and interfere when they meet.
Thomas Young
Time:1807s
Method:A double-slit experiment is an experiment to demonstrate the wave and particle properties of microscopic objects such as photons or electrons.
Experimentally:In the double-slit experiment, particles pass through the slit to form a series of alternating streaks on the screen. These streaks are caused by the interference of particle waves, which interfere with each other when the two slits meet, forming bright and dark streaks.
Experimental conclusion:Yang discovered that light is actually more like a wave than a particle, and the double-slit experiment proved that light can represent the interference properties of waves. This experiment not only shows the wave of microscopic particles, but also reveals the wave-particle duality of particles with both wave and particle properties.
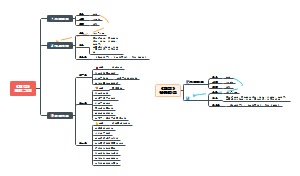
James Clerk Maxwell
Time:1865s
Prophetic electromagnetic wave:Maxwell deduced the existence of electromagnetic waves from his equations,he mathematically derived the speed of electromagnetic waves from his equations and found that it was the same as the speed of light.
Experimental conclusion:Light is a form of electromagnetic wave that reveals the connection between light and electromagnetic phenomena.
Max Planck
Time:1900s
Contribution:Quantum hypothesis
Content
1. The particle nature of light:In the traditional wave theory, light is regarded as a wave phenomenon with wave properties, such as interference and diffraction. However, Planck observed that when solving certain problems, particularly those involving thermal radiation and the photoelectric effect, the fluctuating nature of light could not be satisfactorily explained. Therefore, he proposed the particle nature of light, that is, light consists of a series of discrete, discontinuous particles, namely light quanta (later called photons).
2. Quantization of energy:In Planck's light quantum hypothesis, the energy of photons is not continuous, but discrete and quantized. This means that the energy of light can only be transferred in certain units of energy, namely quanta. This concept of quantization of energy contradicted the continuous energy concept in physics at the time, but was able to successfully explain some experimental phenomena, such as the blackbody radiation law.
Conclusion:The proposed Planck energy quantum hypothesis is of great significance to the development of quantum mechanics. It breaks the continuity assumption of energy in traditional physics, introduces the quantum concept, and finally promotes the establishment of quantum theory. Einstein was influenced by him to propose wave-particle duality.
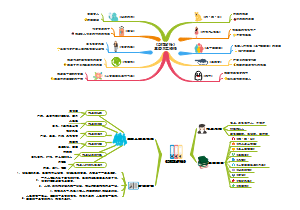
Albert Einstein
Time:1905s
Assumption:Light quantum hypothesis
Content:Light and atomic electrons also have particle properties, light has this particle property called light quantum. Like Planck's energy quantum, the energy of each photon is E=hν, and according to relativistic mass-energy relations, the momentum of each photon is p=E/c=h/λ.
Principle:For a few years after Planck's quantum hypothesis was proposed, it attracted little interest, but Einstein saw its importance. He supported the energy quantum hypothesis, and got an important revelation from it: in the existing physical theory, the object is composed of one atom by one, is discontinuous, and light (electromagnetic wave) is continuous. There is a profound contradiction between the discontinuity of atoms and the continuity of light waves. In order to explain the photoelectric effect, Einstein proposed the light quantum hypothesis on the basis of Planck's energy quantum hypothesis in 1905.