导图社区 CHARACTER
- 32
- 0
- 0
- 举报
CHARACTER
CHARACTER 4&5:Development of Atomic Model、Wave-particle Duality、Atomic Structure
编辑于2022-10-12 18:49:00 北京市- CHARACTER 4&5
- 相似推荐
- 大纲
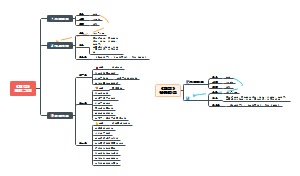
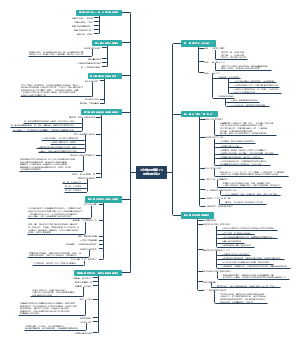
CHARACTER 4&5
Development of Atomic Model
democritus's atomic philosophy
Democritus reasoned that atoms were indivisible anf indestructible.
dalton's atomic theory
All elements are composed of tiny indivisible particles called atoms.
Atoms of the same element are identical. The atoms of апу one element are different from those of any other element.
Atoms of different elements can physically mix together or can chemically combine in simple whole-nuinber ratios to form compounds.
Chemical reactions occur when atoms are separated from each other, joined, or rearranged in a different combination. Atoms of one element, however, are never changed into atoms of another element as a result of a chemical reaction.
J.J. Thomson's plum pudding model
J.J. Thomson's experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles called electrons
the negatively-charged electrons were stuck into a lump of positive charge
Rutherford atomic model
Rutherford's gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus.
the atom is mostly empty space, the positive charge must be localized over a very tiny volume of the atom, which also contains most of the atom's mass.
Rutherford proposed the nuclear model of the atom.
Bohr's model
an electron is found only in specific circular paths, or orbits, around the nuleus
Quantum mechanical model
The quantum mechanical model determines the allowed energies an electron can have and how likely it is to find the electron in various locations around the nucleus of an atom.
Wave-particle Duality
electronmagnetic radiation
light waves(classic physics)
amplitude
The amplitude of a wave is the wave's height from zero to the crest.
wavelength
The wavelength, represented by λ(the Greek letter lambda), is the distance between the crests.
frequency
The frequency, represented by μ(the Greek letter nu), is the number of wave cycles to pass a given point per unit of time.
The units of frequency are usually cycles per second. The SI unit of cycles per second is called the hertz(Hz).
The frequency and wavelength of light waves are inversely related. As the wavelength decreases, the frequency increases.
electronmagnetic spectrum
Electromagnetic radiation includes radio waves, microwaves, infrared visible light, ultraviolet waves, X - rays, and gamma rays. All electromagnetic waves travel in a vacuum at a speed of 2.998×108 m/s.
atomic emission spectrum
When atoms absorb energy, their electrons move to higher energy levels. These electrons lose energy by emitting light when they return to lower energy levels.
when the light emitted by the energized electrons of a gaseous element is passed through a prism,the spectrum consists of a limited number of narrow lines of light. The wavelengths of these spectral lines are characteristic of the element, and they make up the atomic emission spectrum of the element .
the quantum concept and photons
the quantization of energy
In 1900, Planck showed mathematically that the amount of radiant energy(E) of a single quantum absorbed or emitted by a body is proportional to the frequency of radiation(v).
The constant(h), which has a value of 6.626X10^-34 J·s is called Planck's constant. The energy of a quantum equals hv.
The light emitted by an electron moving from a higher to a lower energy level has a frequency directly proportional to the energy change of the electron.
photoelectric effect
electrons are ejected when light shines on a metal
To explain the photoelectric effect, Einstein proposed that light could be described as quanta of energy that behave as if they were particles.
Einstein recognized that there is a threshold value of energy below which the photoelectric effect does not occur. According to E = hv, all the photons in a beam of monochromatic light (light of only one frequency) have the same energy. If the frequency, and therefore the energy, of the photons is too low, then no electrons will be ejected. Only if the frequency of light is above the threshold frequency will the photoelectric effect occur.
Heisenbergr uncertainty principle
The Heisenberg uncertainty principle states that it is impossible to know both velocity and the position of a particle at the same time.
This limitation is critical when dealing with small particles such as electrons, but it does not matter for ordinary-sized objects such as cars or airplanes.
Atomic Structure
Atomic structure
nucleus
proton
found by Eugen Goldstein in 1886
neutron
James Chadwick confirmed the existence of neutron in 1932
most of the mass
electron
discovered by J.J. Thomson in 1897
most of the volume
distinguishing among atoms
atomic number
An element's atomic number is the number of protons in the nucleus of an atom of that element.
mass number
The total number of protons and neutrons in an atom.
isotopes
Isotopes are atoms that have the same number of protons but different numbers of neutrons.
Because isotopes of an element have different numbers of neutrons, they also have different mass numbers.
Despite these differences, isotopes are chemically alike because they have identical numbers of protons and electrons, which are the subatomic particles responsible for chemical behavior.
atomic mass unit
An atomic mass unit (amu) is defined as one twelfth of the mass of a carbon-12 atom.
atomic mass
The atomic mass of an element is a weighted average mass of the atoms in a naturally occurring sample of the element.
To calculate the atomic mass, we must know three things: the number of stable isotopes of the element, the mass of each isotope, and the natural percent abundance of each isotope.
To calculate the atomic mass of an element, multiply the mass of each isotope by its natural abundance, expressed as a decimal, and then add the products.
electron arrangement
electron configuration
In an atom, electrons and the nucleus interact to make the most stable arrangement possible. The ways in which electrons are arranged in various orbitals around the nuclei of atoms are called electron configurations.
the aufbau principle
electrons occupy the orbitals of lowest energy first.
Pauli exclusion principle
An atomic orbital may describe at most two electrons. To occupy the same orbital, two electrons must have opposite spins.
hund's rule
electrons occupy orbitals of the same energy in a way that makes the number of electrons with the same spin direction as large as possible.
energy level
Each possible electron orbit in Bohr’s model has a fixed energy. The fixed energies an electron can have are called energy levels. The energy levels in atoms are unequally spaced, the higher energy levels are closer together.
A quantum of energy is the amount of energy required to move an electron from one energy level to another energy level. The energy of an electron is therefore said to be quantized.
atomic orbital
For each energy level, the Schrödinger equation leads to a mathematical expression, called an atomic orbital, describing the probability of finding an electron at various locations around the nucleus.
The energy levels of electrons in the quantum mechanical model are labeled by principal quantum numbers (n).
Each energy sublevel corresponds to one or more orbitals of different shapes. The orbitals describe where an electron is likely to be found.
c(the spped of light) = wavelength × frequncy