导图社区 SCs-exo在骨形成微环境中的神经代替疗法
- 11
- 0
- 0
- 举报
SCs-exo在骨形成微环境中的神经代替疗法
这是一篇关于A multifunctional neuromodulat的思维导图,主要内容包括:Background,Results,Discussion,Inspiration。
编辑于2025-06-25 23:44:05- 外泌体
- 骨形成
- 施万细胞
- 相似推荐
- 大纲
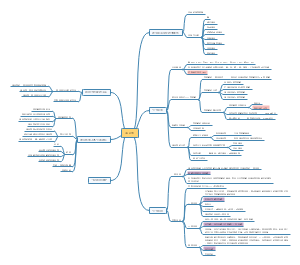
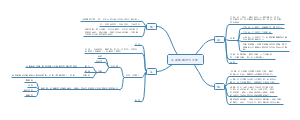
A multifunctional neuromodulation platform utilizing Schwann cell-derived exosomes orchestrates bone microenvironment via immunomodulation, angiogenesis and osteogenesis
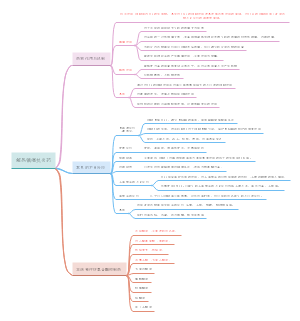
Background
There is a major challenge in the treatment of large bone defects, when the self-repair capability of bone is not enough, the bone substitute materials are promising approaches.
Challenge
The skeleton has long been recognized as a richly innervated tissue. Previous studies have demonstrated that intact innervation is essential for bone healing
SCs(Schwann cells) are crucial for nerve regenberation and functional recovery owing to their role in secreting neuro-trophic factors, supporting the survival of damaged neurons, and promoting axon regrowth,suggesting that SCs may further amplify their synergistic actions on bone healing by promoting innervation in the defect area.
For cell-based therapies still have some intrinsic shortcomings, the cell-free tissue engineering, such as exosomes, has been rapidly developed. Gelatin methacryloyl hydrogel (GelMA) was fabricated to load exosomes to ensure their sustainable and stable release.
the new way to overcome problems
The multifunctional role and mechanism of how SC Exo orchestrate the complex bone healing microenvironment during bone regeneration has not been reported.
The unclear points at present
Results
Characterization of SC Exo and Exo/Hydrogel
Exo/Hydrogel are considered to be beneficial for cell proliferation and nutrient transfer
No significant differences in weight loss between the hydrogel and hydrogel loaded with exosomes
BMSCs could grow and proliferate well on the Exo/Hydrogel system.
The release behavior of exosomes loaded in the hydrogel is consistently great
Early effects on innervation, osteoimmune, and vascular systems trigged by exo/hydrogel in vivo
the expression of CGRP and β3-Tubulin after Exo/Hydrogel treatment increased significantly
M1 macrophages (iNOS-positive cells), while more dominant M2 macrophages (CD206-positive cells) were detected in the Exo/Hydrogel group
Exo/Hydrogel markedly facilitated macrophage polarization toward the M2 phenotype and induced a local anti-inflammatory micro-environment.
Exo/Hydrogel decreased the expression of pro-inflammatory cytokines including TNF-α and IL-1β
immune micro-environment
It was observed that Exo/Hydrogel led to the maximum increase in the number and area of new blood vessels
the formation of blood vessels
Cranial bone regeneration by exo/hydrogel
Exo/Hydrogel-treated animals, more newly formed bone tissues were present in the early bone repair zone, and in the later stages, confluent and dense bony structures were more prominent.
mature and complete osseous bridges were detected in the defect areas of the Exo/Hydrogel group
There is a rapid initiation of bone repair driven by Exo/Hydrogel
The Exo/Hydrogel group demonstrated the highest BV/TV compared with the other two groups throughout the repair process.
The expression of OCN and collagen I gradually decreased in the Exo/Hydrogel group at 12 weeks compared with the control.
The formation rate of new bone is faster
bone formation
Validation of multiple effects of exo/hydrogel in vitro
With the participation of Exo/Hydrogel, M1 markers and pro-inflammatory cytokines (CD86, iNOS, and TNF-α) were significantly inhibited, but the gene expression of M2 markers and anti-inflammatory cytokines (CD206, ARG, and TGF-β3) were considerably elevated when compared with those in the control group
The production of TNF-α and IL-1β in LPS-stimulated macrophages decreased significantly after Exo/Hydrogel treatment
There are significant differences in the number of nodes and segments, as well as in the total length of the vascular networks among different groups
qRT-PCR results showed significant upregulation of proangiogenic genes
The expression of ALP, RUNX2, and OCN in the Exo/Hydrogel group was evidently stronger
Enhancement of osteogenesis was found in BMSCs in the presence of Exo/Hydrogel
Exo/Hydrogel significantly increased the number of migrated BMSCs
Exo/Hydrogel notably enhanced the osteogenesis and migration of BMSCs.
Activation of TGF-β signaling by exo/hydrogel for enhanced BMSCs osteogenesis
The expression levels of key effector genes, including BMP6 and TGF-β1, were profoundly upregulated in the Exo/Hydrogel group
It was known that TGF-β elicits effects on bone formation by activating their receptors to induce phosphorylation of a group of intracellular transcription factors, known as SMADs.
p-SMAD2/3 was shown to intensively co-localize with the nucleus when the cells were stimulated with Exo/Hydrogel, indicating the activation of SMAD2/3
the levels of TGF-β1 and p-SMAD2/3 expression were all upregulated in BMSCs treated with Exo/Hydrogel
TGF-β1/p-SMAD2/3 pathway is activated to promote the osteogenic effect of BMSCS
Discussion
In this study,the application of SCs-exo in the bone formation, a cell-free approach, released with the GelMA hydrogel.
innovation
The combination of GelMA hydrogel and SC Exo represents a convenient and stable approach to develop multifunctional scaffolds
higher stability and more simplified preservation methods than SCs
simultaneous enrichment of many secretory factors from SCs, which can function together
Inspiration
limited mechanical properties are still the major problem of GelMA
most studies involved the non-load bearing sites in animal models and could not represent the actual physiological process of large bone defects repair in clinical therapy.
There is still the need for the extracellular vesicles production to be more convinient and stable