导图社区 nri.2017.7生态失调与免疫系统
- 26
- 0
- 1
- 举报
nri.2017.7生态失调与免疫系统
缺乏Toll样受体(TLR)信号适配器的小鼠骨髓分化一级反应蛋白88(MYD88)----分明的肠道微生物群,粘膜相关细菌数量增加以及细菌向肠系膜淋巴结的易位增加
编辑于2022-03-21 15:29:55- 生态失调
- 先天性淋巴细胞:炎症性疾病的主要参与者
下图是一篇文献领读的思维导图,讲述了先天性淋巴细胞:炎症性疾病的主要参与者,感兴趣的朋友收藏下图了解吧!
- 文献阅读领读--Nature期刊--综述--免疫专栏--第2017.10期
文献阅读领读--Nature期刊--综述--免疫专栏第2017.10期--Neutrophils as protagonists and targets in chronic inflammation.
- nri.2017.7生态失调与免疫系统
缺乏Toll样受体(TLR)信号适配器的小鼠骨髓分化一级反应蛋白88(MYD88)----分明的肠道微生物群,粘膜相关细菌数量增加以及细菌向肠系膜淋巴结的易位增加
nri.2017.7生态失调与免疫系统
社区模板帮助中心,点此进入>>
- 先天性淋巴细胞:炎症性疾病的主要参与者
下图是一篇文献领读的思维导图,讲述了先天性淋巴细胞:炎症性疾病的主要参与者,感兴趣的朋友收藏下图了解吧!
- 文献阅读领读--Nature期刊--综述--免疫专栏--第2017.10期
文献阅读领读--Nature期刊--综述--免疫专栏第2017.10期--Neutrophils as protagonists and targets in chronic inflammation.
- nri.2017.7生态失调与免疫系统
缺乏Toll样受体(TLR)信号适配器的小鼠骨髓分化一级反应蛋白88(MYD88)----分明的肠道微生物群,粘膜相关细菌数量增加以及细菌向肠系膜淋巴结的易位增加
- 相似推荐
- 大纲
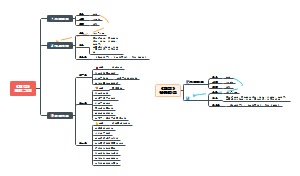
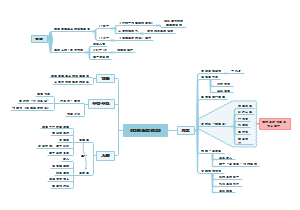
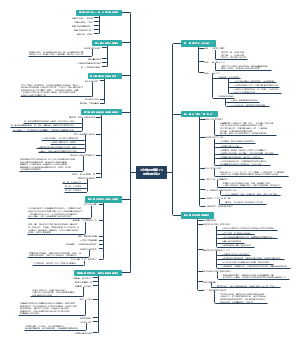
nri.2017.7 Dysbiosis and the immune system
Types of dysbiosis
Bloom of pathobionts
Enterobacteriaceae
Loss of commensals
autism spectrum disorder ----Lactobacillus reuteri, Bacteroides fragilis
Clostridium difficile-induced inflammation----Clostridium scindens
Loss of diversity
alpha diversity
Oringins of dysbiosis
Infection and inflammation
mouse models of infection with Citrobacter rodentium and Salmonella enterica subsp. enterica serovar Typhimurium
Inflammation induced by dextran sodium sulfate or genetic deficiency of interleukin-10 (Il10) in mice
inflammation-induced outgrowth of members of the Enterobacteriaceae family can promote the development of colorectal cancer and sepsis
Diet and xenobiotics
a low-fibre diet reduces microbial diversity in mice
a high-fat diet reduces microbial diversity in mice
antibiotics, sweeteners and dietary emulsifiers
Genetics
A twin study identified the abundance of multiple taxa of the intestinal microbiota influenced by host genetics
Genomic studies have identified an impact of host genetics on colonization with particular taxa in mice
the genetic influence on microbial composition may be involved in the manifestation of certain phenotypes
[eg] Christensenellaceae and low body mass index
Familial transmission
maternal factors alone do not suffice to explain an individual's microbiota assembly
Environmental transmission is additional importance
Other causes
circadian disruption
maternal high-fat diet
pregnancy
physical injury
Immune control of microbial homeostasis
The innate immune system in the regulation of microbial composition
pattern recognition receptors (PRRs)
mice deficient in the Toll-like receptor (TLR) signalling adaptor myeloid differentiation primary response protein 88 (MYD88) ----distinct intestinal microbiota, increased numbers of mucosa-associated bacteria and increased translocation of bacteria to the mesenteric lymph nodes
flagellin sensor TLR5(-/-) ----alteration of intestinal microbiota leads to the manifestation of hyperphagia, metabolic syndrome, high levels of Enterobacteriaceae
Myd88(−/−)mice
NOD-like receptors (NLRs)
nucleotide-binding oligomerization domain-containing protein 1 (NOD1)(-/-)----bacterial population is expanded (Clostridiales, Bacteroidesspp., segmented filamentous bacteria (SFB) and Enterobacteriaceae)
Nod2(−/−)mice---- increased burden of commensals, increased proportion of mucosa-associated bacteria
human NOD2 deficiency
a failed study----well-designed experimental controls are important
NOD-, LRR- and pyrin domain-containing 6 (NLRP6) inflammasome----activating caspase 1, ASC speck formation, maintenaning of a stable microbial community in the intestine
deficiency of ASC and caspase 1----dysbiosis
α-defensins(-/-)---- normal bacterial numbers, altered microbial composition
antimicrobial lectin REGIIIγ (by Paneth cells ): separating the microbiota and the epithelial surface
RORγt+ ILCs ( innate lymphoid cells)----producing IL-22
T-bet+ ILCs----producing interferon-γ (IFNγ), tumour necrosis factor (TNF); regulating the composition of the microbiota
The adaptive immune system in the regulation of microbial composition
B cells
secretory IgA
T follicular helper (TFH) cells
programmed cell death protein 1 (PD1)----Regulating Bacterial communities and IgA production
Invariant natural killer T (iNKT) cells
responding to microbial glycolipids
intraepithelial lymphocytes
expressing γδ T cell receptors
Impact of dysbiosis on the host immune system
Signalling to innate immunity
microbial cell components
different immunogenicity of LPS---stimulating TLR4, activating nuclear factor-κB and tolerating endotoxin
TLR2
metabolites
the bile acid conjugate taurine, the amino acid histamine and the polyamine spermine----influencing NLRP6 activity----the secretion of IL-18
IL-22
Signalling to adaptive immune cells
degradation of secretory IgA
Dysbiosis and immunological diseases
IBD
Coeliac disease
Rheumatoid arthritis
T1D
Asthma
Multiple sclerosis
Dysbiosis in diagnostics and therapy
Diagnostics
DNA sequencing for the identification of strains and their genomes, RNA sequencing for the determination of microbial gene activity, metabolome analysis
dysbiosis index
mucosal samples> luminal faecal samples
shotgun metagenomic sequencing, non-targeted metabolomics, microbial biomarkers
the diagnostic value of the microbiome may lie in differentiating subtypes of IBD that share common clinical symptoms as well as IBD and other intestinal inflammatory diseases
the microbiome as a diagnostic tool reaches far beyond the intestine
Parkinson disease
Alzheimer disease
therapy
faecal microbiota transplantation (FMT)
antibiotics
administration of probiotics
dietary prebiotics----personalized diets
a shift in the composition of the intestinal microbiota may have positive effects on the host: anticancer drug cyclophosphamide alters the intestinal microbial community----potentiating the anticancer effect of the drug
Challenges and future avenues
[1] appropriate controls for definiting for a microbial ecosystem as dysbiosis
[2] the concept of dysbiosis deserves a functional rather than a taxonomic interpretation
[3] the extent and manifestation of dysbiosis seem to be highly context dependent. (particular microbial configuration----particular genomic mutation on disease susceptibilty, susceptibility to dysbiosis----host genotype and environmental microbial repertoire)
[4] insights into the molecular mechanisms that drive dysbiosis-associated pathologies----'probiotic' and 'postbiotics' intervention
Abstract: In this Review, we categorize dysbiosis in conceptual terms and provide an overview of immunological associations; the causes and consequences of bacterial dysbiosis, and their involvement in the molecular aetiology of common diseases; and implications for the rational design of new therapeutic approaches. A molecular-level understanding of the origins of dysbiosis, its endogenous and environmental regulatory processes, and its downstream effects may enable us to develop microbiome-targeting therapies for a multitude of common immune-mediated diseases. ([1]changes in lifestyle and environmental factors>gene evolution----abnormal microbiome's composition and function, termed dysbiosis----focusing on bacterial of the gastrointestinal tract----common multifactorial diseases. [2]the healthy intestinal microbial community's characters. [3]the basic definition of dysbiosis is limited:interindividual variability, vertical transmission, housing effects, variations in pathogen screening in animal facilities and other factors, adaptations of the microbiome to altered environmental conditions or changes in the state of the host may be benificial or neutral or harmful----the use of a narrow definition of dysbiosis, namely statistically and functionally.)